Tibial Plateau Co28Cr6Mo Overview
What This Tibial Plateau Co28Cr6Mo Implant Is
Our Tibial Plateau Co28Cr6Mo implant is a high-strength cobalt‑chrome‑molybdenum (CoCrMo) tibial tray designed for demanding knee reconstruction cases. It serves as a load‑bearing tibial baseplate or reconstruction tray in:
- Complex tibial plateau fracture fixation
- Primary and revision total knee arthroplasty (TKA)
- Post‑traumatic and segmental tibial plateau defects
We use UNS R31538 / ASTM F75 cobalt alloy, engineered for durable, wear‑resistant support under repeated high joint loads.
Who This System Is For
We built this system for teams that need predictable performance and streamlined logistics:
- Orthopedic surgeons specializing in trauma and knee arthroplasty
- Hospitals and surgery centers needing reliable, standardized trays
- OEMs and implant brands seeking ASTM F75 CoCrMo tibial baseplates as private‑label components
- Distributors looking for a modular tibial plateau CoCrMo system for both trauma and arthroplasty lines
Why Co28Cr6Mo for the Tibial Plateau
Using Co28Cr6Mo cobalt chrome on the tibial side gives you:
- High strength and stiffness for comminuted, depressed, or osteoporotic plateau fractures
- Excellent wear resistance against cobalt chrome femoral components in TKA
- Corrosion resistance in the synovial environment, supporting long‑term performance
- Biocompatible cobalt chromium with low ion release when manufactured and finished to current standards
The result is a high‑strength cobalt tibial prosthesis that tolerates aggressive loading and early mobilization.
Typical Clinical Use and Indications
You’ll typically consider this CoCrMo tibial plateau component in:
- Schatzker I–VI tibial plateau fractures, especially high‑energy, multi‑fragment patterns
- Knee fracture reconstruction where joint surface restoration and load sharing are critical
- Primary TKA in active or high‑BMI patients needing a wear‑resistant Co28Cr6Mo knee implant
- Revision TKA where bone stock is limited and a stable tibial baseplate is essential
- Trauma and post‑traumatic arthritis requiring a combined fixation–arthroplasty approach
Across these scenarios, the goal is the same: stable fixation, reliable load transfer, and long‑term, wear‑resistant joint function in the tibial plateau.
Tibial Plateau Co28Cr6Mo Material Properties
Co28Cr6Mo Alloy Composition & Standards
Our Tibial Plateau Co28Cr6Mo implant uses a high‑purity cobalt‑chromium‑molybdenum alloy that meets UNS R31538 and fully complies with ASTM F75 for cast surgical implants.
Design Features of Tibial Plateau Co28Cr6Mo Implants
Our Tibial Plateau Co28Cr6Mo implants are built to give surgeons predictable mechanics, strong fixation, and smooth articulation in demanding U.S. knee cases.
Anatomical Contouring & Dual-Condyle Geometry
We match native tibial anatomy to help you restore joint line and stability:
- Side-specific medial/lateral contours to respect natural asymmetry
- Dual‑condyle geometry for stable femoral tracking and even load transfer
- Low-profile edges to reduce soft‑tissue irritation and impingement
| Feature | Clinical Advantage |
|---|---|
| Anatomic footprint | Better coverage, less overhang |
| Dual‑condyle design | Stable contact through full ROM |
| Low-profile rim | Less soft tissue conflict |
Posterior Slope, Articular Surface & Load Distribution
We keep the mechanics simple and reliable:
- Built‑in posterior slope options to match your TKA philosophy
- Optimized articular surface curvature for smooth motion and reduced peak stresses
- Broad load‑sharing platform to limit subsidence in osteoporotic or post‑trauma bone
| Design Element | Purpose |
|---|---|
| Posterior slope | Support flexion and ligament balance |
| Curved articular surface | Reduce contact stress and wear |
| Wide baseplate | Improve load distribution |
Porous Coating & Surface Finish Options
You can choose fixation strategy based on bone quality and surgeon preference:
- Porous coated zones for cementless osseointegration in suitable bone
- Polished or matte finishes on non‑articulating regions to manage soft tissue interaction
- Surface treatments compatible with advanced surface engineering workflows similar to our orthopedic surface treatment services.
| Surface Option | Use Case |
|---|---|
| Porous coated | Biological fixation / younger patients |
| Matte base | Cemented technique, reduced glare |
| Polished posts/edges | Smooth soft‑tissue interface |
Modular Design, Screw Holes, Stems & Augments
We build in flexibility for complex trauma and revision work:
- Modular stems (short/long) for diaphyseal support
- Locking and non‑locking screw holes to secure fragments or grafts
- Metal augments (wedges/blocks) to handle bone loss and depression
- Unified instrument and tray system to keep OR workflow fast and familiar
| Modular Option | Typical U.S. Use Case |
|---|---|
| Offset/straight stems | Revision and poor metaphyseal support |
| Locking screw holes | Comminuted plateau fractures (Schatzker V–VI) |
| Augments | Post‑traumatic defects and revision TKA |
Clinical Indications for Tibial Plateau Co28Cr6Mo
Tibial plateau fractures (Schatzker I–VI)
For complex tibial plateau fractures, including Schatzker I–VI patterns, a Tibial Plateau Co28Cr6Mo implant gives you a rigid, stable platform when standard plates or screws aren’t enough. It’s especially helpful when:
- The articular surface is comminuted or depressed
- There’s metaphyseal bone loss or segmental defects
- You need a load‑sharing “reconstruction tray” to support grafts, blocks, or augments
In high‑demand trauma centers, we see it used as a definitive solution after high‑energy injuries where restoring joint congruity and early motion is critical.
Primary and revision total knee arthroplasty
A Co28Cr6Mo tibial plateau (tibial tray/baseplate) is a solid choice for:
- Primary TKA in active patients where wear resistance and long‑term stability are priorities
- Revision TKA with bone loss, failed tibial components, or loosening where stronger support and stemmed options are required
The high‑strength cobalt chrome molybdenum platform is designed to pair with standard femoral components and polyethylene inserts, similar to our cobalt‑chromium knee implant components.
High‑energy trauma and osteoporotic bone
In the US, we see two key groups benefit most:
- Younger patients with high‑energy trauma (falls from height, MVAs) needing strong, fatigue‑resistant support
- Older patients with osteoporotic bone, where a rigid CoCrMo tibial tray spreads load across a larger surface and works well with stems, augments, and bone graft
This combination helps reduce subsidence risk and maintains alignment under heavy cyclical loading.
Patient selection and contraindications
Good candidates for a Tibial Plateau Co28Cr6Mo implant usually:
- Can commit to rehab and follow weight‑bearing protocols
- Have adequate soft‑tissue coverage around the knee
- Need durable fixation for complex fracture or arthroplasty scenarios
You should be cautious or avoid use when:
- There’s a known or suspected cobalt/chromium alloy allergy or metal hypersensitivity
- Active infection is present in or around the joint
- Bone stock is so poor that even stemmed, augmented fixation can’t achieve stability
- The patient cannot tolerate surgery or expected rehab load
Careful imaging, lab work, and history (especially prior metal reactions or failed implants) are key before committing to a cobalt chrome molybdenum tibial plateau solution.
Tibial Plateau Co28Cr6Mo in Surgical Techniques
Our Tibial Plateau Co28Cr6Mo implants are built to support both traditional ORIF and modern minimally invasive knee approaches. The cobalt chrome molybdenum construction gives you a stable, low‑profile platform that works well with locking screws, adjunct plates, and modular stems, whether you’re dealing with high‑energy trauma or complex reconstruction.
ORIF and minimally invasive compatibility
- Compatible with standard ORIF workflows, including lateral and anterolateral approaches
- Low‑bulk geometry designed to fit through smaller incisions in minimally invasive setups
- Works with common locking and cortical screw systems for stable metaphyseal fixation
- Ideal for staged or hybrid trauma–arthroplasty strategies in comminuted tibial plateau fractures
Preoperative planning and imaging
- Rely on CT with 3D recon to map articular depression, split patterns, and posteromedial fragments
- Use templating to match tibial plateau Co28Cr6Mo sizes, planned posterior slope, and stem length
- Confirm implant position vs. existing hardware or prior implants before going to the OR
- For custom or complex cases, we can support advanced planning similar to how we handle our titanium alloy orthopedic components used in trauma and joint workups
Intraoperative alignment, reduction, and fixation
- Restore mechanical axis using intramedullary or extramedullary guides tied to the tibial tubercle and ankle center
- Rebuild the joint surface first: elevate depressed fragments, support with bone graft or wedges, then lock in with the tibial plateau Co28Cr6Mo tray
- Use the dual‑condyle design to check medial–lateral balance and rotational alignment against the femoral condyles
- Lock screws subchondrally in a raft pattern to buttress the plateau and reduce the risk of collapse
Postoperative rehab and weight‑bearing
- Early CPM or active ROM is encouraged if fixation is stable, focusing on extension first, then flexion
- Typical protocol in the U.S. market:
- 0–6 weeks: toe‑touch or partial weight‑bearing depending on fracture pattern and bone quality
- 6–12 weeks: advance to full weight‑bearing as radiographic healing appears solid
- For arthroplasty‑type reconstructions with robust metaphyseal support, earlier weight‑bearing may be appropriate based on surgeon judgment
- Close follow‑up with serial X‑rays to monitor for subsidence, loss of reduction, and hardware integrity
Technical Specifications of Tibial Plateau Co28Cr6Mo
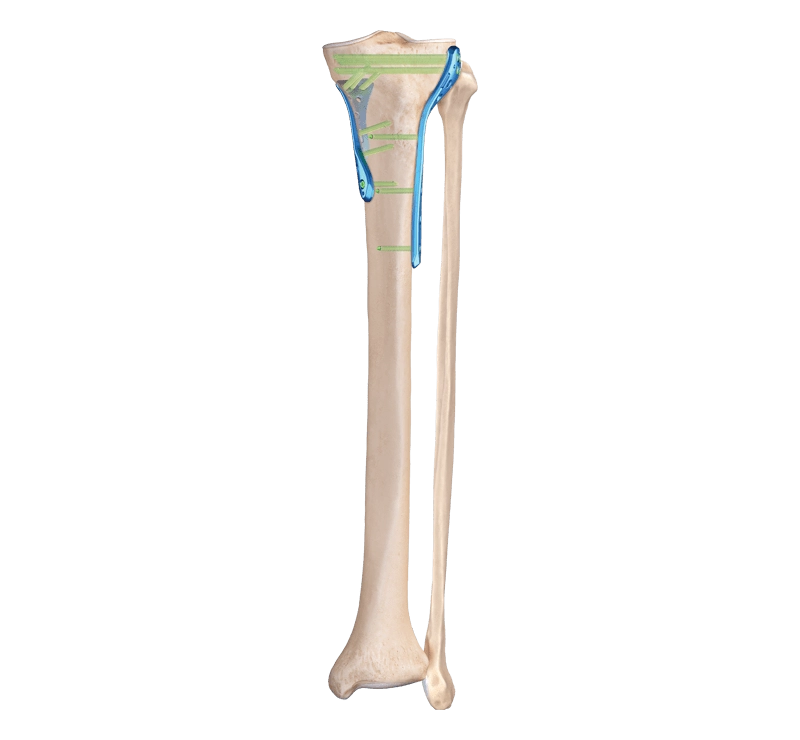
Size and Dimensional Catalog
We standardize our Co28Cr6Mo tibial plateau sizes so surgeons can mix, match, and plan quickly:
| Parameter | Range (Typical) |
Performance and Clinical Outcomes of Tibial Plateau Co28Cr6Mo
Tibial plateau Co28Cr6Mo implants have a strong track record in U.S. practice, especially where durability and high load demand are non‑negotiable.
Long‑term survivorship and revision rates
In both trauma reconstruction and total knee arthroplasty (TKA), Co28Cr6Mo tibial plateau components consistently show:
- High 10–15+ year survivorship, especially when alignment and fixation are correct.
- Lower revision rates in active and higher‑BMI patients compared with softer alloys, thanks to the alloy’s strength and wear resistance.
For hospital systems and OEMs, that translates into fewer returns, fewer reoperations, and more predictable outcomes.
Clinical scores and functional recovery
Patients treated with a cobalt chrome molybdenum tibial plateau component typically achieve:
- Improved KSS, WOMAC, and KOOS scores once rehab milestones are met.
- Reliable early weight‑bearing when fixation and bone quality allow, helping patients get back to daily activities and work faster.
Surgeons tend to see smoother recovery curves in high‑demand knees because the construct doesn’t “give” under load.
Wear performance vs titanium and other materials
Compared with titanium tibial trays and other alloys, a wear‑resistant Co28Cr6Mo knee implant offers:
- Lower polyethylene wear rates under high contact stress.
- Better scratch and abrasion resistance, which is critical when there’s any risk of third‑body particles.
In load‑intensive environments—similar to aerospace and energy components that demand high hardness and precision, like a front diffuser with high pressure recovery and durability—this alloy’s performance edge is obvious.
Risk profile: subsidence, loosening, metallosis
With proper indication and technique, the risk profile is favorable:
- Subsidence: Low when used with adequate metaphyseal support and, where needed, stems or augments.
- Aseptic loosening: Reduced by stable fixation and appropriate cement or porous‑coated designs that promote bone in‑growth.
- Metallosis / ion release: Rare in modern Co28Cr6Mo systems, especially with clean counterface design and stable articulation; still, it’s important to screen for cobalt/chromium sensitivity in at‑risk patients.
For U.S. surgeons, that balance—high strength, low wear, and a manageable risk profile—makes tibial plateau Co28Cr6Mo a dependable choice in both complex fractures and demanding TKA cases.
Regulatory and Quality Standards for Tibial Plateau Co28Cr6Mo
Our Tibial Plateau Co28Cr6Mo implants are built to meet strict U.S. and international requirements so surgeons and hospitals can plug them into existing workflows with confidence.
Regulatory clearances
- Designed for FDA 510(k) pathways in the U.S. and CE-marked configurations for the EU (where applicable by model).
- Configurable documentation sets to support local market registrations and hospital value analysis committees.
- Full technical files, risk analysis, and validation data are compiled under a controlled quality system.
ISO-based quality and biocompatibility
- Manufactured under an ISO 13485–compliant quality management system, with validated processes and documented controls.
- Materials and components are tested and documented to support ISO 10993 biocompatibility expectations for cobalt chrome molybdenum tibial trays.
- We back this with robust material and process verification through independent and in‑house testing, supported by our own testing and quality assurance procedures.
Traceability, lot control, and documentation
- Every Tibial plateau Co28Cr6Mo implant carries full lot traceability from raw UNS R31538 alloy to finished sterile device.
- We maintain:
- Heat and melt records
- Manufacturing batch histories
- Inspection and test reports
- Standard documentation packs include:
- IFU and surgical labeling
Customization Options for Tibial Plateau Co28Cr6Mo
I treat every Tibial plateau Co28Cr6Mo project as a custom engineering job, not a one‑size‑fits‑all part. For complex trauma, revisions, or unusual anatomy, customization is often the difference between “acceptable” and “excellent” outcomes.
Patient-Specific & 3D Printed Tibial Plateau Options
We support fully patient‑specific Co28Cr6Mo tibial plateau components based on CT or MRI data:
- Custom trays for bone defects (segmental loss, malunions, post‑tumor resections)
- Asymmetric geometries for varus/valgus deformities
- Built‑in augments and buttresses to match actual defect shape
- 3D printed trial models to confirm fit before final production
A patient‑matched tibial plateau Co28Cr6Mo implant lets you preserve more native bone and improve load transfer across the joint.
Surface Customization: Polished, Matte, Porous
You can define the surface strategy for each case
| Region | Surface Option | Typical Use |
|---|---|---|
| Articular / bearing areas | High polish | Low wear vs femoral component |
| Peripheral non‑articular | Matte | Reduced glare, easier orientation intraop |
| Bone contact / defect filling | Porous / rough | Enhanced osseointegration, cementless fixation |
We control finish through precision casting and machining processes similar to our high‑accuracy investment casting workflows, then apply orthopedic‑grade polishing and surface treatments.
Size and Geometry Tailoring for Complex Defects
For high‑energy fractures, revisions, and post‑traumatic deformities, we routinely tailor:
- Medial / lateral width and depth
- Posterior slope and coronal alignment
- Offset stems and stepped augments
- Buttress wings for posteromedial or posterolateral support
This lets you match the true defect and restore joint line and slope more accurately than with off‑the‑shelf trays.
Scan-to-Implant Workflow
We keep the workflow straightforward for U.S. hospitals, OEMs, and trauma centers:
- Submit imaging
- Upload DICOM CT (knee + proximal tibia) via secure portal
- Optional: share planned resection or reconstruction template
- Digital planning & design review
- We generate a 3D model and preliminary tibial plateau Co28Cr6Mo design
- You receive a PDF + 3D viewer file with key dimensions and planned fixation points
- Surgeon approval
- You confirm size, slope, stem options, porous regions, and screw hole layout
- Revisions are turned around quickly until you sign off
- Manufacture & QC
- Component is produced using controlled casting/machining similar to our precision metal part manufacturing capabilities
- 100% inspection, lot traceability, and documented material certs
- Delivery & case support
- Sterile or non‑sterile options (per agreement)
- Preoperative sizing sheet and implant map included for OR efficiency
This scan‑to‑implant model lets you keep control over the reconstruction strategy while we handle the heavy lifting on design and manufacturing of the Co28Cr6Mo tibial plateau.
Ordering Tibial Plateau Co28Cr6Mo Components

Ordering codes & configuration options
I keep the Tibial plateau Co28Cr6Mo system simple to order and easy to stock.
You can configure each Co28Cr6Mo tibial plateau implant by:
- Base code – tibial size, medial/lateral width, right/left, and thickness
- Fixation options – keel vs stemmed, screw-hole patterns, modular augments
- Surface options – polished or matte articular surface, porous-coated undersurface
- Set type – trauma tibial plateau reconstruction tray or TKA tibial baseplate kit
A single order sheet or digital form covers all options, so your team can standardize codes across hospitals and OEM programs. For OEMs, I can match your internal part-number logic and labeling.
Pricing models & bulk purchasing programs
For U.S. hospitals, ASC groups, and distributors, I support:
- Tiered volume pricing for system-wide adoption
- Bundled trauma or TKA tray pricing to simplify budgeting
- OEM / private-label pricing for long-term platform partnerships
If you’re already sourcing cobalt-chrome joint implants, I can align pricing with your existing cobalt chrome lines, including our chromium-cobalt-molybdenum hip joint machining service for full lower-limb portfolios.
Lead times, shipping & regional availability
I build around fast turnaround and predictable supply:
- Standard sizes: usually ready to ship from U.S.-served stock
- High-volume configs: scheduled production with locked capacity
- Custom or OEM runs: lead time agreed in advance with firm delivery windows
I ship nationwide across the United States with tracked, temperature‑controlled packaging when required, and can coordinate direct-to-hospital, distributor hub, or OEM warehouse deliveries.
Support for tenders, OEM supply & private label
I actively support:
- Hospital and IDN tenders – full technical documentation, samples, and value files
- OEM supply – neutral branding, engineering collaboration, and stable long-term contracts
- Private label programs – your logo, your IFU, our Co28Cr6Mo tibial plateau manufacturing
My team can help with pricing files, technical data packs, and validation documents so you can move quickly on bids and new product lines.
Support, Training, and Resources for Tibial Plateau Co28Cr6Mo Users
I back every Tibial plateau Co28Cr6Mo implant with hands-on support so your team can move fast and stay safe in the OR.
Surgical Technique Guides & Training
- Step-by-step surgical technique PDFs (ORIF, trauma, TKA revision)
- On-site or remote in-service training for surgeons, fellows, and OR staff
- Case-based workshops using real U.S. trauma and arthroplasty scenarios
- Standardized sets and workflows to shorten the learning curve
Digital Tools: Planning, Sizing, Video
- Pre-op planning templates and printable sizing charts
- Short, focused surgical videos for key steps and “tricky” scenarios
- Digital technique files and catalogs that plug into your existing workflow
- Access to online resources through our product and precision casting services pages
Clinical Evidence & Reference Library
- Clinical data packets: survivorship, functional scores, and wear data
- U.S.-relevant case series for tibial plateau fractures and knee reconstruction
- Quick-reference cards for indications, contraindications, and implant selection
- Evidence summaries formatted for hospital committees and value analysis teams
Technical Support & Account Management
- Dedicated technical support for sizing, compatibility, and OR troubleshooting
- Fast-response phone/email support for urgent surgical questions
- Assigned account manager for quotes, trials, and standardization projects
- Full product documentation and traceability through our orthopedic product catalog



Reviews
There are no reviews yet.