When you choose medical cobalt-based alloy artificial joint castings, you’re balancing three things: fatigue strength, wear resistance, and regulatory safety. I design and supply CoCrMo alloy castings specifically for orthopedic implant manufacturers and medical OEMs that have to hit tight performance targets and strict standards in the US and global markets.
What Buyers Are Really Looking For
Most engineers and sourcing teams searching for CoCrMo alloy castings or ASTM F75 artificial joints are trying to solve one of these problems:
- Need reliable, repeatable cobalt-chrome orthopedic implants that pass validation and regulatory review
- Need a qualified investment casting partner that understands medical-grade requirements, not just industrial casting
- Need clear technical data: chemistry, mechanical properties, and biocompatibility proof, not marketing claims
My focus is on medical grade CoCr castings for:
- Orthopedic implant manufacturers
- Medical device OEMs
- Contract manufacturers building hip, knee, shoulder, and ankle systems for major brands
Role in Artificial Joint Systems
Cobalt-chrome (CoCrMo) castings are typically used in the high-load, articulating components of joint replacement systems, where failure is not an option:
- Femoral condyle casting for total knee arthroplasty
- Femoral neck casting and hip prosthesis CoCrMo components for total hip systems
- Acetabular cup cobalt alloy and double-jointed cup designs for demanding hip motion
- Ankle joint prosthesis alloy parts for smaller but highly loaded joints
In these positions, biocompatible cobalt-based alloy provides:
- High wear resistance against UHMWPE, ceramics, and other counterfaces
- Excellent corrosion resistance in vivo, even in aggressive physiological environments
- High fatigue strength for long-term load-bearing cycles
My role as a supplier is to provide vacuum melted cobalt alloy castings that integrate cleanly into your orthopedic joint replacement components, with the consistency, documentation, and quality systems your regulatory and engineering teams expect.
Material specs for medical cobalt-based alloy artificial joint castings

For our medical cobalt-based alloy artificial joint castings, I use a standard CoCrMo chemistry based on Co-28Cr-6Mo that aligns with ASTM F75 for cast cobalt-chromium surgical implants. Typical controlled ranges include:
- Cobalt (Co) – balance
- Chromium (Cr) – ~27–30% for passive corrosion protection
- Molybdenum (Mo) – ~5–7% for solid-solution strengthening
- Low carbon, controlled nickel, manganese, silicon, iron, and impurities per ASTM F75 limits
This gives you a stable, repeatable alloy that passes medical-grade requirements, not just generic casting specs you’ll see in a basic casting alloy guide.
Mechanical properties for CoCrMo joint castings
I design these CoCrMo alloy castings to meet or exceed ASTM F75 mechanical properties after proper heat treatment and processing:
- Ultimate tensile strength (UTS): typically ≥ 655 MPa
- Yield strength (0.2% offset): typically ≥ 450 MPa
- Elongation: usually ≥ 8–10% to avoid brittle behavior
- Fatigue performance: high fatigue strength under cyclic loading, tuned for hip, knee, and ankle joint prosthesis applications
We run full mechanical testing per heat/batch so orthopedic OEMs get traceable data for qualification and regulatory files.
Biocompatibility, corrosion, and wear performance
This medical-grade CoCrMo alloy is well established in hip and knee prosthesis parts for the U.S. market thanks to its in‑vivo stability:
- Biocompatibility: proven clinical history in cobalt-chrome orthopedic implants; low ion release when processed correctly
- Corrosion resistance: strong passive film in chloride-containing body fluids, resisting pitting and crevice corrosion
- Wear performance: excellent wear resistance against UHMWPE, ceramics, and other CoCrMo surfaces, supporting long-term THA and TKA service life
For femoral condyle, femoral neck, double-jointed acetabular cup, and ankle joint components, this combination of chemistry, mechanical strength, and in‑body wear behavior is exactly what I optimize for long-term implant performance.
Product types and artificial joint components
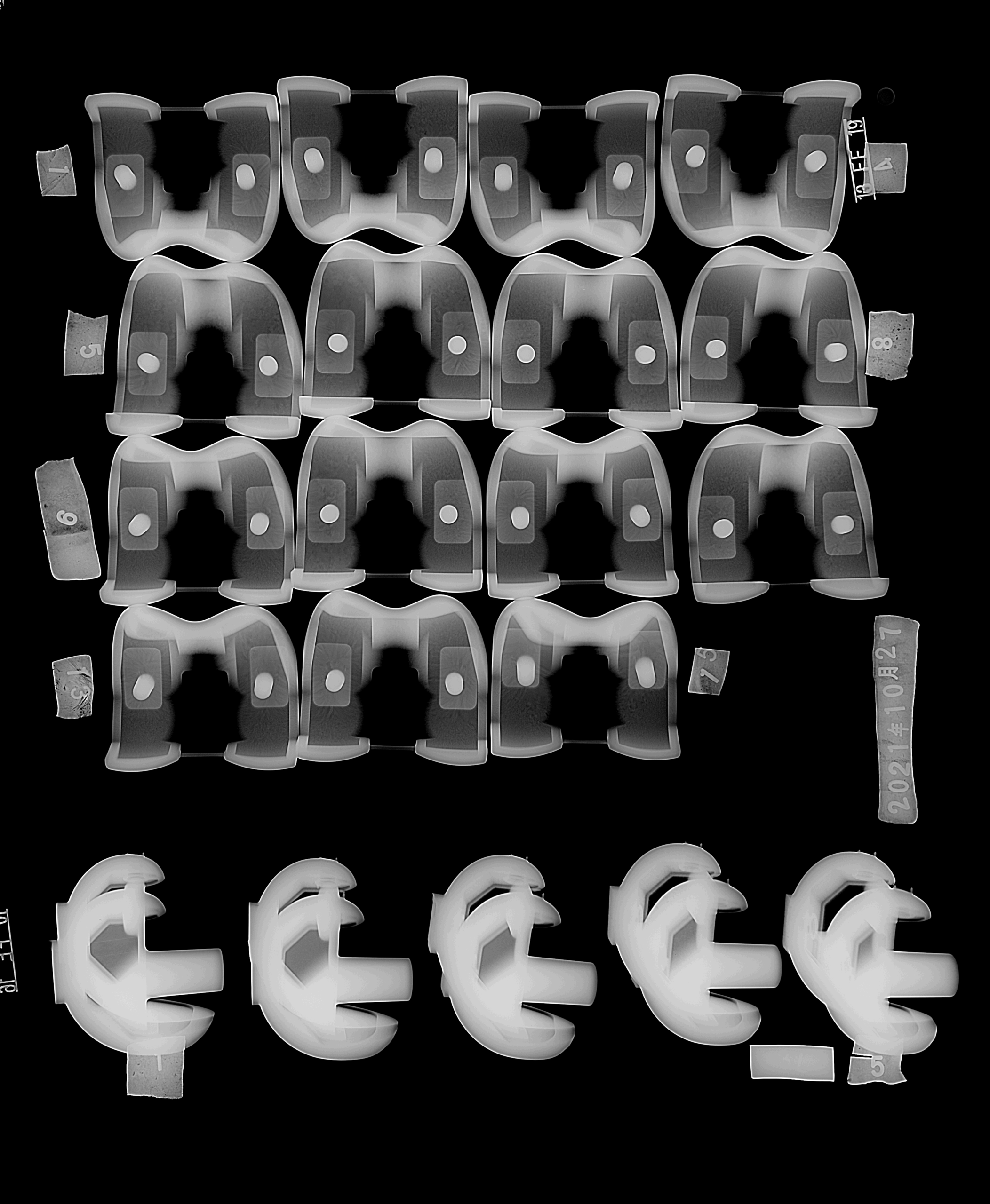
Femoral condyle cobalt alloy castings
We supply precision femoral condyle CoCrMo alloy castings for knee joint implants, with smooth articular surfaces designed for ultra-low wear against UHMWPE inserts. Our medical cobalt-based alloy artificial joint castings are produced by investment casting for tight tolerances, consistent radii, and stable fixation features that match your instrument sets and cementing techniques.
Femoral neck and hip prosthesis CoCrMo parts
For hip systems, we provide femoral necks, heads, and proximal bodies in medical-grade cobalt-chrome-molybdenum. These hip prosthesis CoCrMo parts are engineered for high fatigue strength, stable taper connections, and long-term corrosion resistance in modular THA designs. We can match your existing stem geometries or help optimize new profiles for U.S. patient anatomy.
Double-jointed acetabular cups and hip socket components
We cast double-jointed acetabular cups and hip socket components with precise inner geometry for liners and locking mechanisms. Our cobalt-chrome orthopedic implants deliver hard, wear-resistant bearing surfaces for metal-on-poly or metal-on-ceramic constructs, and we support custom hole patterns, anti-rotation features, and porous or roughened regions ready for coating.
Ankle joint prosthesis cobalt-based alloy castings
For ankle replacements, we produce talar and tibial CoCrMo alloy castings with complex contours and stable fixation platforms. These ankle joint prosthesis alloy components focus on smooth articulation, resistance to edge loading, and reliable performance under demanding gait cycles typical of active U.S. patients.
Custom cobalt-chrome components for multiple joints
Beyond standard hip and knee, we support custom cobalt-chrome components for shoulder, elbow, small joint, and trauma-adjacent applications. Whether you need a new femoral condyle casting family, a unique hip geometry, or matched sets for multi-joint systems, we offer OEM-grade engineering support, including integration with our chromium-cobalt-molybdenum machining service for hip joint components to deliver near net-shape or fully finished parts.
Manufacturing Process for Medical Cobalt-Based Alloy Artificial Joint Castings
Precision investment casting for complex CoCrMo implants

For our medical cobalt-based alloy artificial joint castings, we rely on precision investment casting (lost-wax casting) to hit tight tolerances and complex geometries like femoral condyles, femoral necks, double-jointed cups, and ankle joint components. Wax patterns, ceramic shells, and controlled burnout let us produce thin sections, smooth contours, and undercuts that would be costly or impossible with traditional machining alone. You can see a similar approach in our broader precision casting services, adapted here specifically for medical CoCrMo alloys.
Vacuum melting, pouring, and solidification control
We use vacuum melting and controlled pouring to keep gas, inclusions, and contamination to a minimum—critical for cobalt-chrome orthopedic implants. Key controls include:
- Strict alloy charge management and clean raw materials
- Vacuum or inert atmosphere melting for consistent chemistry
- Controlled mold preheat and pour temperature for sound filling
- Managed cooling rates to refine microstructure and reduce porosity
This gives us stable CoCrMo alloy properties from heat to heat, ready for ASTM F75–compliant applications.
Dimensional accuracy and machining allowance
Our tooling and process control are built around medical OEM expectations in the U.S.:
- High dimensional accuracy right out of the mold
- Near-net-shape castings to cut machining time and cost
- Small, predictable machining allowances on critical joint surfaces (articulating areas, taper interfaces, locking features)
This lets your team focus machining only where needed—tapers, critical fits, and final joint surfaces—while keeping per-part cost under control.
Surface treatment, polishing, and finishing
To get parts “implant-ready,” we run a dedicated finishing workflow for medical grade CoCrMo:
- Gate and riser removal, controlled grinding, and deburring
- Shot blasting or micro-blasting for uniform baseline surfaces
- Multi-step mechanical polishing up to mirror finish on articulating zones
- Optional passivation or cleaning steps matched to your downstream sterilization
The result is a smooth, low-roughness surface that supports wear resistance and corrosion performance in vivo.
Non-destructive testing and dimensional inspection
Every batch of medical cobalt-based alloy castings goes through a defined NDT and inspection flow:
- X-ray or CT to check for internal porosity and shrink
- Dye penetrant testing for surface-breaking defects
- Full dimensional inspection (CMM, gauges, and custom fixtures) against your CAD and drawing tolerances
We document all results for traceability and validation. For more detail on how we control each step upstream, you can reference our broader manufacturing process overview, which underpins our medical casting lines as well.
Applications in Orthopedic Joint Replacement
Total hip arthroplasty (THA) cobalt-based components
In total hip arthroplasty, our medical cobalt-based alloy artificial joint castings are used for femoral heads, femoral necks, and acetabular cups. CoCrMo’s high wear resistance and fatigue strength make it ideal for hard-on-poly and hard-on-hard bearing surfaces, especially for active U.S. patients who expect long implant life and high mobility.
Total knee arthroplasty (TKA) femoral and tibial castings
For TKA, we produce femoral condyle castings and matching tibial components in medical grade CoCrMo. The smooth, polished cobalt-chrome surface reduces wear against UHMWPE inserts and supports stable kinematics under repeated loading. You can see how this applies in real designs with our precise anatomical femoral condyle implant components.
Shoulder and ankle joint replacement alloys
We supply cobalt-chrome castings for shoulder humeral heads, glenoid-side components, and ankle joint prosthesis parts. These applications demand strong, corrosion-resistant alloys that can handle complex motion patterns without loosening or excessive wear, which is exactly where CoCrMo excels.
Modular and custom orthopedic prosthesis systems
Our CoCrMo alloy castings are engineered for modular stems, necks, cones, and custom revision or tumor prosthesis systems. We support OEMs with:
- Tight-tolerance mating surfaces for modular junctions
- Custom geometries for complex reconstructions
- Consistent alloy chemistry and mechanical performance across product lines
This gives U.S. orthopedic manufacturers the flexibility to design patient-matched and modular systems while maintaining reliable, scalable CoCrMo casting supply.
Quality Assurance for Medical Cobalt-Based Alloy Artificial Joint Castings
When you buy medical cobalt-based alloy artificial joint castings from us, you’re getting parts built under a tight, medical-grade quality system designed for U.S. orthopedic OEMs.
ISO 13485 Medical Quality System
We run production under an ISO 13485–aligned quality management framework for medical device components, including:
- Documented process control from wax pattern to finished CoCrMo casting
- IQ/OQ/PQ for critical equipment and processes
- Full lot-level control to support your device DMR/DHR requirements
For additional metal material categories, we apply similar controls to our high-performance alloy and stainless steel products, so the approach is proven and scalable.
Full Heat and Batch Traceability
Every cobalt-based alloy heat and batch is traceable:
- Unique heat number tied to melt, pour, and inspection records
- Traceability from raw material to final machined casting
- Long-term digital record retention for audits and FDA submissions
You always know exactly which heat went into which femoral condyle, femoral neck, double-jointed cup, or ankle joint component.
Mechanical, Chemical, and Metallurgical Testing
We back our CoCrMo alloy castings with complete test data:
- Chemical analysis to ASTM F75 and customer specs
- Mechanical testing: tensile, yield, elongation, hardness, and fatigue on representative samples
- Metallurgical evaluation: microstructure, porosity, inclusion rating, and grain size
Third-party or customer-witnessed testing is available, and we can align with your internal validation plans. More on our testing discipline can be seen in our dedicated testing and quality workflow.
Regulatory and Documentation Support for OEMs
We make it easier for your regulatory and quality teams to do their jobs:
- Mill test reports (MTRs), certificates of conformance (CoC), and full test reports
- Material and process documentation to support FDA 510(k), PMA, and EU MDR submissions
- Support for PPAP, FAI, and custom control plans for critical joint replacement components
You get medical grade CoCrMo alloy castings with the documentation, traceability, and quality backbone needed for serious orthopedic brands in the U.S. market.
Key benefits of medical cobalt-based alloy artificial joint castings
Wear and corrosion resistance that lasts
Our medical cobalt-based alloy artificial joint castings are built for the long haul. Compared with stainless steel or many titanium-on-poly bearing pairs, CoCrMo castings deliver:
- Outstanding wear resistance in high-contact zones like femoral condyle, femoral neck, and double-jointed acetabular cups
- High corrosion resistance in body fluids, cutting down on metal ion release and extending implant life
- Stable performance under repetitive motion in hip, knee, and ankle joints
For OEMs focused on long-term clinical performance, this is a proven, low-risk material choice.
High fatigue strength for real-world loads
Cobalt-chrome (CoCrMo) shines under cyclic loading, which matters in every step, twist, or climb your patient takes:
- High fatigue strength for heavy and active patients
- Excellent crack resistance in thin-wall and complex geometries
- Reliable performance for decades of use with proper design and fixation
This makes our medical grade CoCr castings ideal for critical load-bearing parts like hip prosthesis necks and knee joint implant castings.
Design flexibility and OEM customization
We support orthopedic OEMs that need both performance and freedom of design:
- Complex, near-net-shape castings for femoral condyles, acetabular cups, and ankle joint prosthesis parts
- Custom cobalt-chrome components for hip, knee, shoulder, and small joints
- Tight tolerances and controlled machining allowance to match your CAD and surgical technique
If you’re developing a new system or optimizing an existing platform, we partner with your engineers from concept through validation, similar to how we support custom metal projects in other sectors like precision alloy components.
Reliable supply and scalable capacity
For U.S. orthopedic manufacturers, consistency and delivery are just as important as metallurgy:
- Stable, vacuum-melted cobalt-based alloy supply with controlled heats and traceability
- Scalable production to support both pilot runs and global implant platforms
- Export-ready logistics and documentation aligned with medical device market needs
You get a dependable OEM partner that can grow with your product line and support long-term programs in hip, knee, and ankle reconstruction.
Technical data, documentation, and OEM support
We back every medical cobalt-based alloy artificial joint casting with hard data and clear documentation so your regulatory and engineering teams get what they need fast.
CoCrMo material datasheets & technical parameters
We provide full technical data for all CoCrMo alloy castings (including ASTM F75 cobalt-chrome):
| Item | Typical Data (Co-28Cr-6Mo) |
|---|---|
| Standard | ASTM F75 / ISO 5832-4 |
| Condition | Vacuum-melted investment casting |
| Ultimate tensile strength | 655–900 MPa (95–130 ksi) |
| Yield strength (0.2%) | 450–690 MPa (65–100 ksi) |
| Elongation | ≥ 8–12% |
| Hardness | 25–35 HRC (customizable by heat treat) |
| Density | ~8.3 g/cm³ |
You get:
- Material datasheets (PDF)
- Chemical composition reports per heat
- Mechanical property test summaries
- Corrosion and wear test data on request
CAD models & design support for CoCrMo implants
For femoral condyle, femoral neck, double-jointed acetabular cups, and ankle joint prosthesis castings, we support your design teams with:
- 3D CAD files (STEP, IGES, Parasolid) of our standard casting envelopes
- Recommended casting draft, radii, and wall thickness guidelines
- DFM feedback to reduce machining time and cost
- Early-stage design reviews for new hip and knee prosthesis platforms
Our team is used to working with US-based engineering groups that need quick design iterations and clear tolerance stacks.
Case studies, performance & clinical reliability
We share evidence-based data to help your regulatory, marketing, and clinical teams:
- Wear and fatigue test reports for CoCrMo hip and knee components
- Case summaries of long-term performance in THA and TKA systems
- Comparative data versus other alloys (e.g., stainless, Ti alloys)
- Process capability data (Cp/Cpk) for critical implant dimensions
For buyers who source other metals as well, we can align our material reporting with your existing systems for alloys like those used in high-performance bronze components.
OEM partnership, customization & engineering collaboration
We operate as a long-term OEM partner, not just a casting supplier:
- Custom tooling & fixtures for your proprietary hip, knee, shoulder, and ankle systems
- Co-development of new CoCrMo components with joint design and casting feasibility in mind
- Dedicated project engineer for each major OEM platform
- NDA-backed collaboration for design, process windows, and cost-down projects
You get fast response times, controlled change management, and documentation ready for FDA submissions and ISO 13485 audits.
FAQ: Medical cobalt-based alloy artificial joint castings
How long do CoCrMo artificial joint castings last in the body?
Our medical cobalt-based alloy artificial joint castings are based on Co-28Cr-6Mo (ASTM F75) and engineered for long-term load-bearing use. In properly designed systems and normal clinical use, they’re targeted for 15–25+ years of service, with high fatigue strength and excellent wear resistance against UHMWPE and ceramic counterfaces.
How do cobalt-based alloy castings perform vs. other implant materials?
- Higher wear resistance than most stainless steels and many titanium-on-poly combinations.
- Excellent corrosion resistance in blood and synovial fluid environments.
- High fatigue and tensile strength, ideal for femoral condyle, femoral neck, double-jointed acetabular cups, and ankle joint prosthesis components.
What customization options and design limits can you support?
We support full OEM customization for hip, knee, shoulder, and ankle prosthesis parts:
- Custom geometries for femoral condyle, femoral neck, and acetabular cup cobalt alloy castings
- Modular interfaces, fixation features, and surface prep for coatings
- Design review for wall thickness, radii, and gating-friendly geometry
We typically review CAD early to confirm: - Castability and shrinkage control
- Dimensional tolerance targets
- Machining allowance and surface finish requirements
What are your typical MOQs for medical CoCrMo castings?
- New custom designs: usually start around 50–100 pcs per size, depending on complexity and tooling.
- Mature or high-volume parts: we scale into the thousands of pieces per year per model.
For specialized or developmental projects, we can discuss lower pilot volumes alongside tooling amortization.
What are your lead times and production capacity?
Typical timelines (after drawing/tooling sign-off):
- Tooling + first article samples (FOT/PPAP): about 6–10 weeks
- Standard production runs: about 4–8 weeks, depending on batch size and quality protocol
We run vacuum-melted investment casting lines with scalable capacity for global orthopedic OEMs. For broader alloy casting capabilities and process info, you can review our medical and industrial casting alloy services.
Can you support global shipping and logistics?
Yes. We routinely ship cobalt-based hip and knee prosthesis parts to North America, Europe, and Asia. We work with:
- FDA-aligned documentation expectations for U.S. OEMs
- Export packing, labeling, and palletization suited for cleanroom-bound components
- Flexible INCOTERMS (FOB, CIF, DAP) depending on your purchasing setup
Are your medical cobalt-based alloy castings biocompatible?
Yes. Our CoCrMo alloy is produced to ASTM F75 with vacuum melting and tight chemistry control to support biocompatibility. Final device biocompatibility (ISO 10993, etc.) is the OEM’s responsibility, but our cast materials and processes are designed specifically for implant applications.
What certifications and standards do you work under?
- Quality system aligned with ISO 13485 medical device requirements
- All heats traceable with full mill certs and heat numbers
- Mechanical and chemical test reports available for each batch (tensile, hardness, microstructure, chemistry)
We also support OEMs with documentation packages to help with FDA and other regulatory filings.
What documentation do you provide with CoCrMo joint castings?
On request we can provide:
- Material certificates (ASTM F75 compliance, chemistry, mechanicals)
- Dimensional inspection reports and NDT records
- Process and lot traceability data, aligned with your DMR/DHF needs
How do I start a custom orthopedic casting project with you?
Send us:
- 3D CAD (STEP/IGES) and 2D drawings with tolerances
- Target material spec (ASTM F75 CoCrMo) and surface finish
- Expected annual volume and launch timeline
We’ll come back with:
- Design-for-casting feedback
- Estimated tooling cost and per-piece pricing
- Lead time, MOQ, and quality/validation plan tailored to your program.


Reviews
There are no reviews yet.